ITACONIC ACID
A replacement for petroleum-based acrylic acid, used to make absorbent materials for nappies; and resins used in high-performance marine and automotive components.
Factfile
Name: Itaconic acid
Synonyms: Methylenesuccinic acid, 2-methylene butanedioic acid
CAS Number: 97-65-4
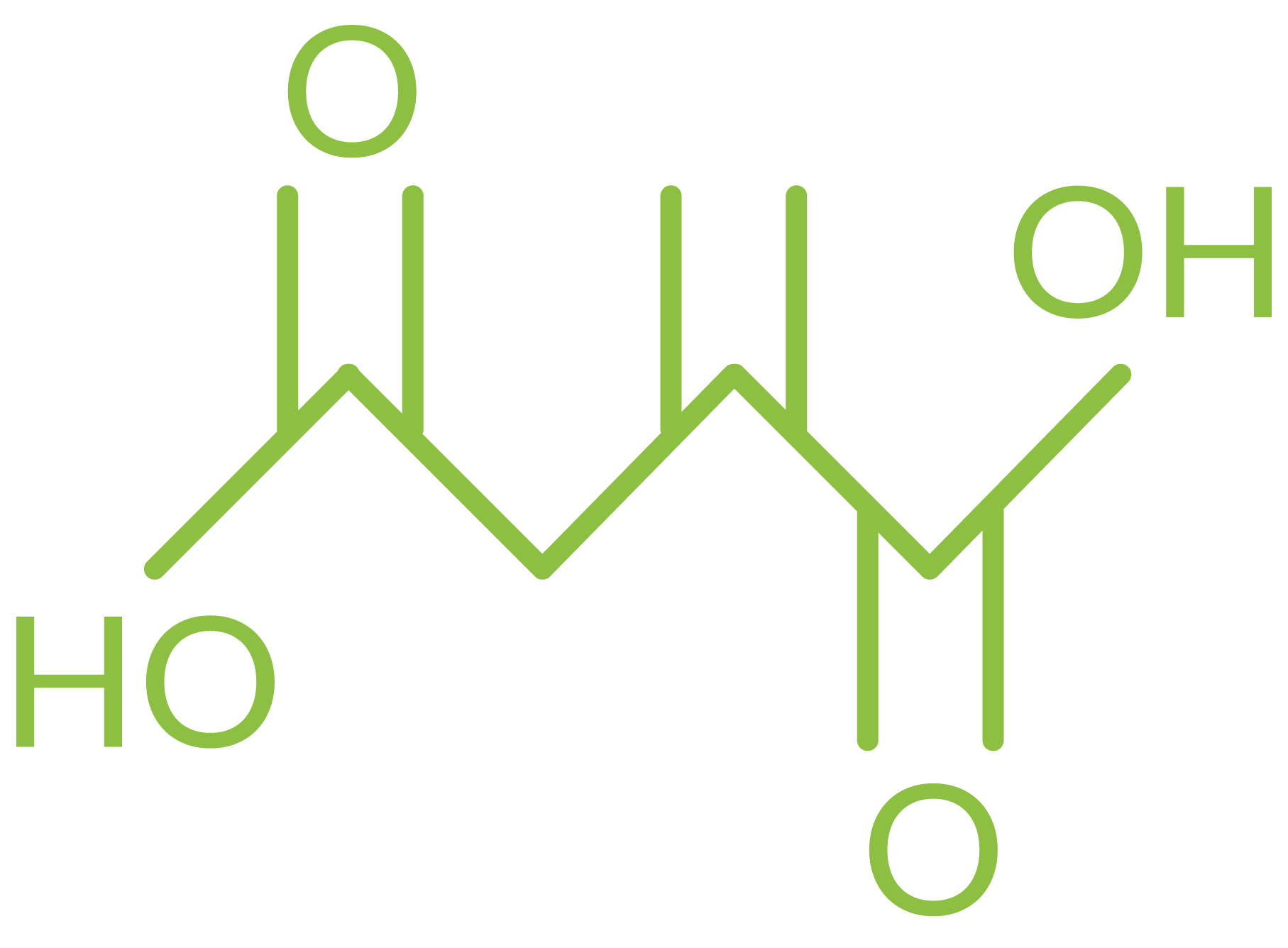
Molecular formula: C5H6O4
MW: 130.10 g mol-1
Patents related to synthesis: 27
Why is it of interest?
Itaconic acid (IA) is a bio-derived platform molecule with a long history, first being synthesised from citric acid in 1837. It is already being produced at scale industrially , with global production of 41 kton (capacity to produce 80 kton) in 2011, none of which is petro-chemically derived. The two carboxylic acid groups can easily be functionalised to give esters and amides, or if reacted with diols or diamines to give polyesters and polyamides. The presence of the alkene allows post polymerisation modification. Additionally the alkene can itself be functionalised or due to activation by the adjacent carboxylic acid, be polymerised via radical initiation similar to petro-chemically derived acrylates and methacrylates.
Feedstocks
All early work regarding IA synthesis utilised citric acid as feedsock to chemocatalytically give the desired product via decarboxylation. This route gives IA in poor yield and selectivity, and the methodology is energy intensive, requiring high pressure and temperature. Other synthetic routes have also been reported and patented although none are economically viable other than biosynthesis. Microbial biosynthetic production of IA utilises the most abundant sugar, glucose, as feedstock. Free sugars xylose, sucrose, mannose and arabinose have successfully been used to produce IA, in addition to starch, sugar beet/sugar cane molasses, jatropha seed cake, olive waste and waste fruit (banana and apple). More complex lignocellulosic feedstocks have been hydrolysed to give sugars for use in IA production (corn cobs, hydrolysed hemicellulose, beech wood hydrolysate) although yields reported have been poor at best, through to nothing if lignin is still present in the system.
Applications

As IA is already produced at scale, there are a number of existing markets where it is employed, the largest of which is as a co-monomer styrene butadiene rubber (SBR) where it increases tack/adhesion in certain applications, such as in coatings. Similarly, itacoinc acid has been employed in emulsion polymerisation for paints and coatings to improve adhesion and wear of the final product. The homopolymer polyitaconic acid, or polyacrylates with a high itaconic acid co-monomer content, have a very high water absorbency leading to applications as super absorbent materials. The other major field of application is as a chelating (metal capture) agent in home and personal care markets. Additionally there are a number of niche applications, including drug delivery in vaccine loaded hydrogels and replacement surfactants.
THE TOP TEN
